By A Mystery Man Writer

Standard Operating Procedure (SOP) and Guideline for the Receipt, Storage, Preparation, Growth Promotion Test, use, and Disposal of microbiological media.
Culture media

EU GMP Annex 1

Microbiological Culture Media: A Complete Guide for Pharmaceutical and Healthcare Manufacturers

Validation of Microbiological Methods

Pharmaceutical Microbiology Resources: Microbiological Culture

The Ultimate Guide to Good Laboratory Practices (GLP)
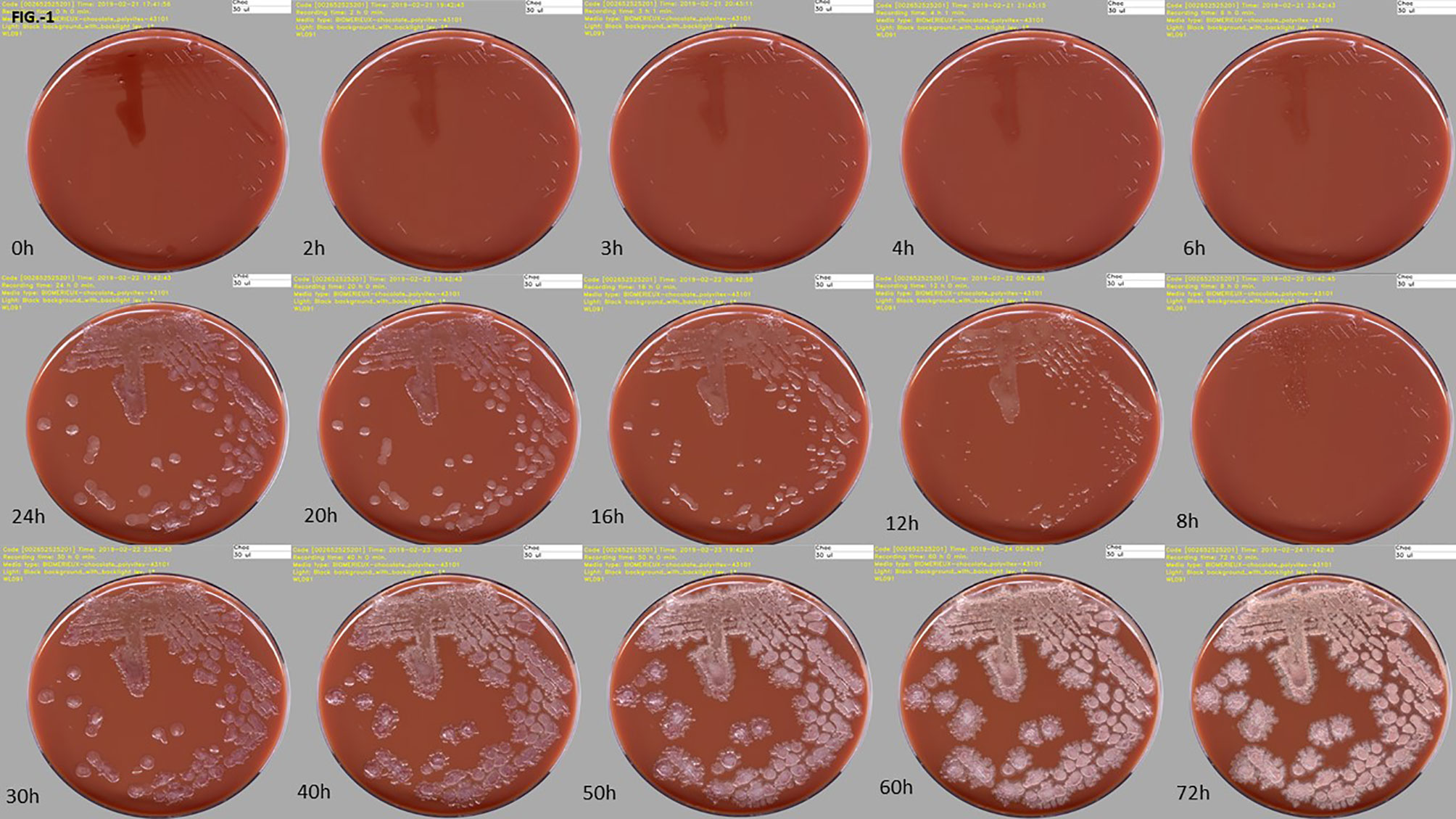
Frontiers Total Laboratory Automation for Rapid Detection and Identification of Microorganisms and Their Antimicrobial Resistance Profiles
Animal Cell Culture Guide
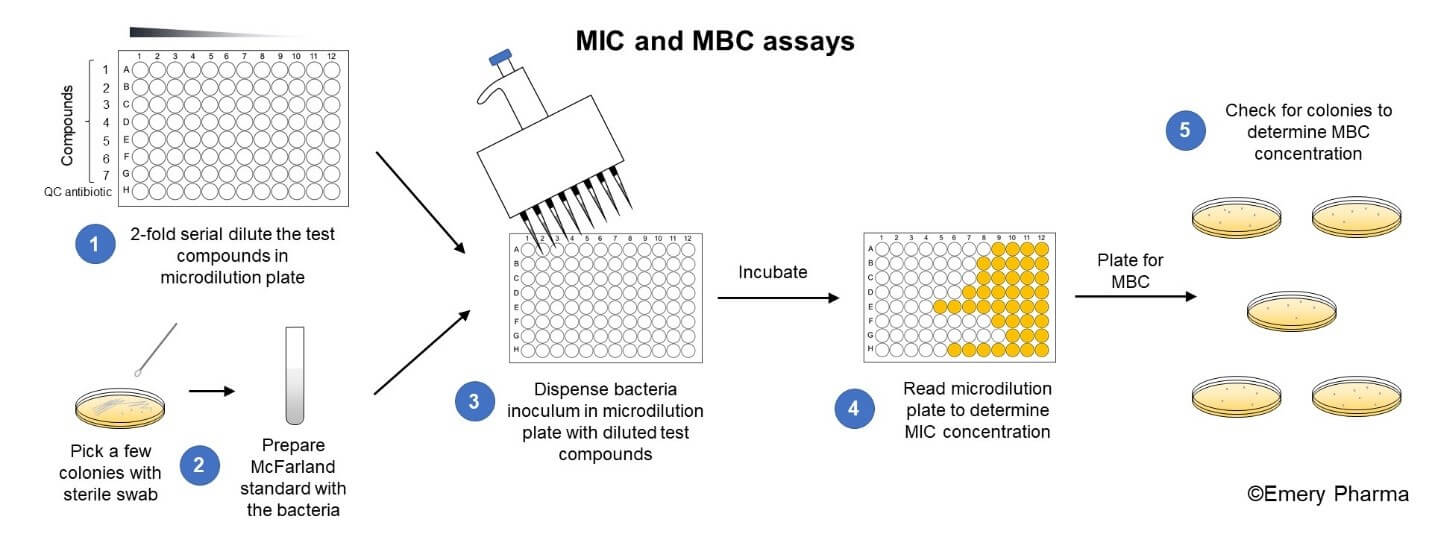
Minimum Inhibitory Concentration (MIC) - Emery Pharma

EN ISO 11133 and Water for the Preparation and Performance Testing

Culture media